|
Mendeleev's Periodic Law states that physical and chemical properties of elements are a periodic function of their atomic masses. 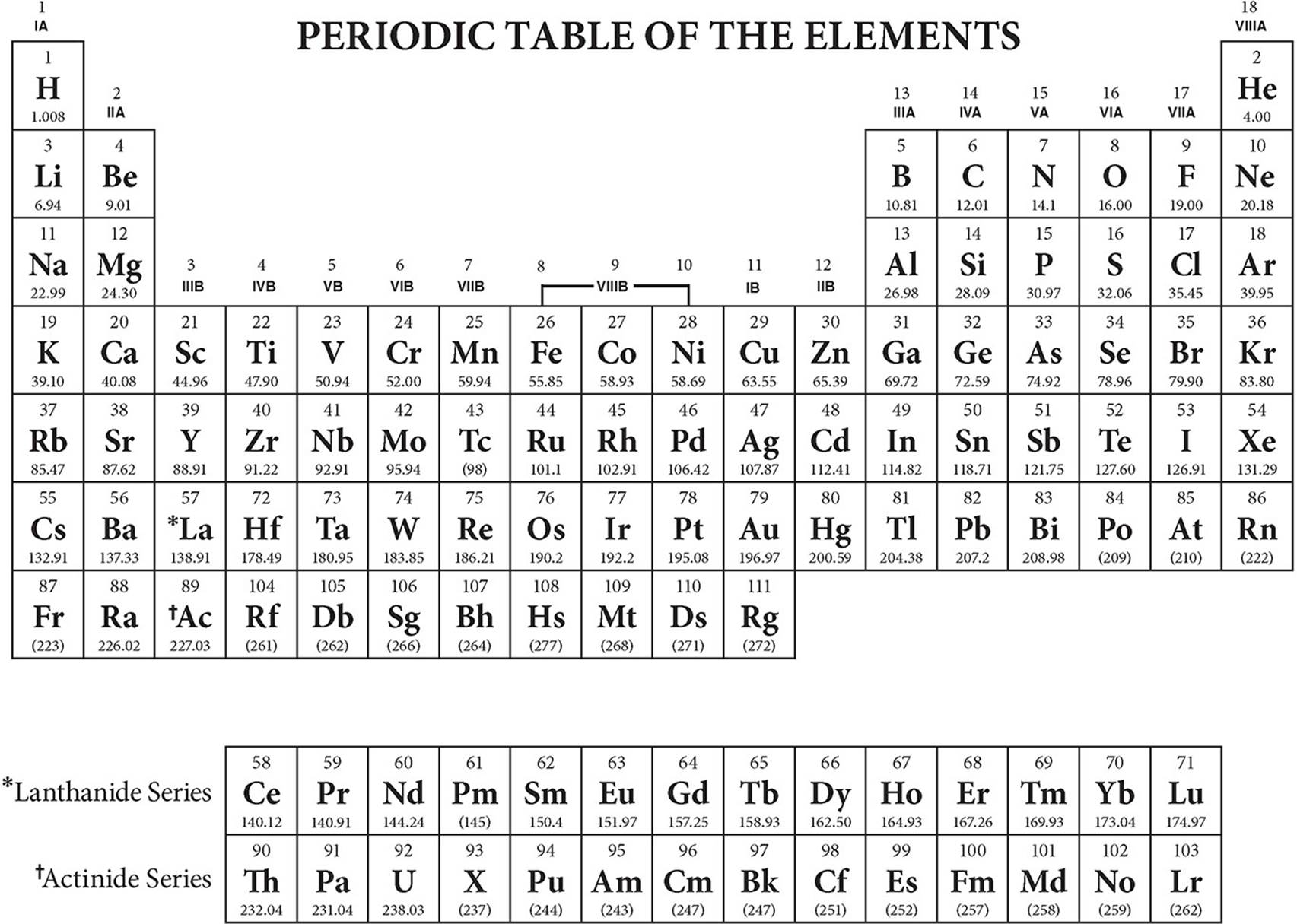
(a) Mendeleev classified elements according to Mendeleev's Periodic Law. (b) Mendeleev's contributions to the concept of a periodic table laid the foundation for the Modern Periodic Table. (a) What was Mendeleev's basis for classification of elements? Similar properties of calcium, strontium and barium were: The elements calcium, strontium and barium were put in one group or family on the basis of their similar properties. These metals impart colour to the flame.They have one electron in the outermost shell, so they form unipositive ions.Similar properties of lithium, sodium and potassium were: Lithium, sodium and potassium elements were put in one group on the basis of their similar properties. From this, we conclude that Dobereiner's triads also exist in the columns of Newland's Octaves. We also know that lithium, sodium, and potassium form a Dobereiner's triad having similar chemical properties. This means that according to Newland's law of octaves, the elements lithium, sodium, and potassium should have similar chemical properties. If we start with lithium as the 1st element, then the 8th element from it is sodium, and according to Newlands' law of octaves, the properties of the 8th element, sodium should be similar to those of the 1st element, lithium.Īgain, if we take sodium as the 1st element, then the 8th element from it is potassium, and according to Newland's law of octaves, the properties of the 8th element, potassium should be similar to those of the 1st element, sodium. Yes, Dobereiner's triads also exist in the columns of Newland's octaves.Įxample: Consider the elements lithium (Li), sodium (Na), and potassium (K) present in second column of Newland's classification of elements. Iron, which resembles cobalt and nickel in properties has been placed far away from these elements.ĭid Dobereiner's triads also exist in the columns of Newland's Octaves? Compare and find out.Newland adjusted two elements Cobalt (Co) and Nickel (Ni) in the same slot, and these were placed in the same column as fluorine, chlorine and bromine which have very different properties than these elements.As more and more elements were discovered, they could not be fitted into Newland's Octaves. This classification did not work with heavier elements i.e., those lying beyond Calcium.Reasons for discarding the Newland's law of Octaves: Why was the law discarded?Īccording to Newland's law of octaves, when elements are arranged by increasing atomic mass, the properties of every eighth element starting from any element are a repetition of the properties of the starting element. The law did not fully apply even within the same family.Įxplain 'Newland's law of Octaves'.Dobereiner failed to arrange all the then known elements in the form of triads.(b) Reasons for discarding Dobereiner's triads: (a) When the atomic mass of A = 7, and that of C is = 39 then according to Dobereiner's triad, atomic mass of B should be mean value of atomic masses of A and C.ħ + 39 2 \dfrac 2 46 = 23 (b) Why were Dobereiner's triads discarded? 
If the atomic mass of A is 7 and that of C is 39, what should be the atomic mass of B? (a) A, B and C are the elements of a Dobereiner's triad. What was the basis of the earliest attempts made for classification and grouping of elements?Įarliest attempts or classification and grouping of elements were based on factors such as: It helps in defining the relationship of one element with another.It helps in correlating the properties of elements with the fundamental properties of all states of matter.It helps in studying the elements in an organized manner.Reasons for classification of elements are: 
What is the need for classification of elements?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
